The batch I pulled out of the fridge last February was the one that finally made me stop guessing and start testing. Eighteen hours at 38°F, 1:8 ratio, medium-coarse on the Comandante, Ethiopian Yirgacheffe from a roaster I trust. On paper, everything was right. In the glass, it tasted like someone had wrung out a wet newspaper and called it coffee.
I’d been making cold brew for about four years at that point, and I’d had off batches before. But this one was different — flat, slightly bitter on the back end, none of the fruit-forward brightness that particular bean is known for. I adjusted the grind coarser. I extended the steep to 24 hours. I tried a different ratio. The bitterness got worse with time, and the coarser grind just made it thinner.
What I hadn’t tested was the water.

My tap water in the Denver metro area runs around 110 ppm total hardness — calcium carbonate equivalent — with bicarbonate alkalinity sitting at roughly 70 ppm. I’d known this in the abstract, the way you know your city’s water report exists without actually reading it. When I finally pulled up the Denver Water quality report and ran my own test with a cheap API hardness kit, the numbers confirmed what I’d been ignoring for years.
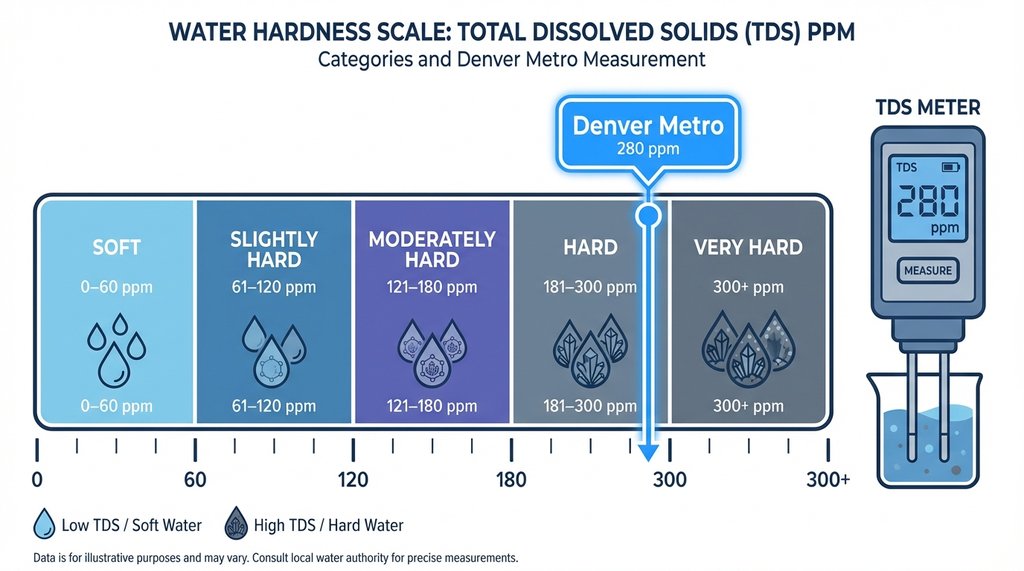
The problem isn’t hardness in the way most people think about it.
Here’s where the standard advice breaks down: most cold brew guides tell you to “use filtered water” and leave it there. The implication is that a Brita pitcher or a PUR faucet filter is sufficient. It isn’t, and the reason is specific enough to be worth understanding.
Activated carbon filtration — which is what Brita Standard cartridges primarily use — is excellent at removing chlorine, chloramine, and volatile organic compounds. It does essentially nothing for dissolved mineral hardness. My tap water at 110 ppm came out of a Brita at 100 ppm. That 10 ppm reduction is within the margin of error for my test kit. For practical purposes, the Brita did nothing to address the actual problem. If you live in an area with hard water, you might even consider researching the best coffee makers for hard water.

The actual problem is bicarbonate alkalinity, not hardness per se.
Calcium and magnesium ions affect extraction differently, and this is where most write-ups get it wrong by treating “hardness” as a single variable. Magnesium actually enhances the solubility of certain flavor compounds — specifically, it improves extraction of aromatic acids and some of the sweeter, more volatile compounds that define a good cold brew’s character. The SCA’s water chemistry research has documented this, and it’s why water with some magnesium hardness (around 10–30 ppm Mg²⁺) tends to produce better-tasting coffee than water with zero mineral content.
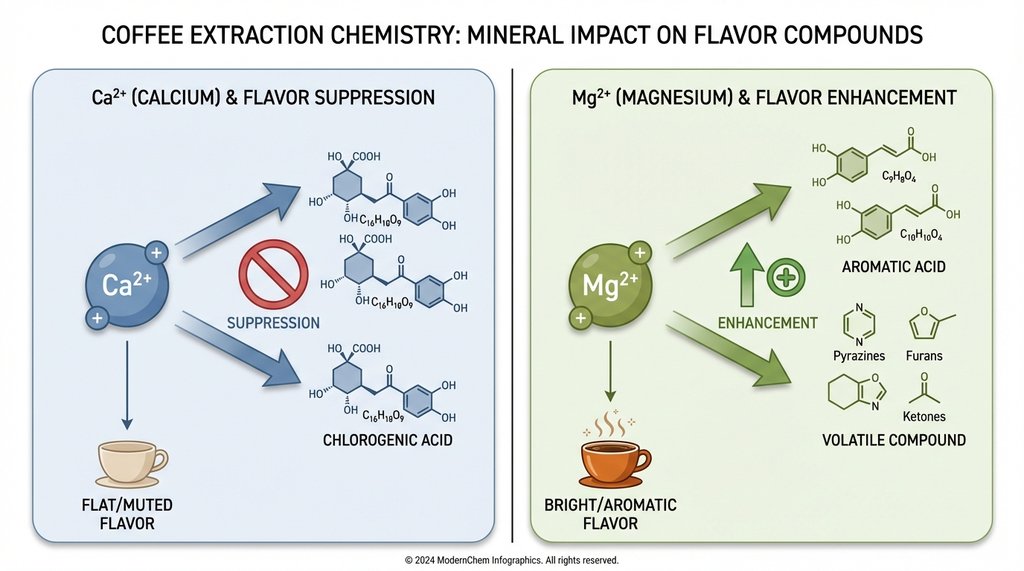
Calcium is more complicated. Calcium ions bind to different flavor compounds, which emphasizes heavier body and can mask delicate, bright notes. But the bigger issue with calcium-heavy hard water is what it brings along: bicarbonate.
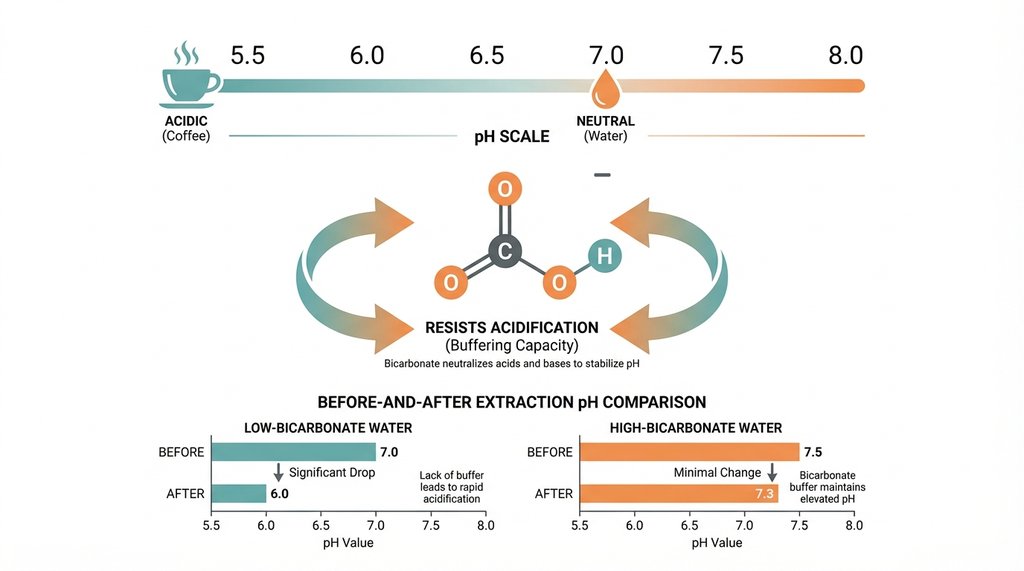
Bicarbonate (HCO₃⁻) is a pH buffer. When your brew water has 70 ppm bicarbonate, it’s actively resisting the acidification that happens during extraction. Coffee extraction is partly a pH-dependent process — as you extract, the water becomes


4 thoughts on “How Hard Water Ruins Cold Brew & The Simple Filter Fix”