The recall notice sat in my inbox at 6:47 AM on a Tuesday in February, forwarded from a client running a regional grocery chain in the Mid-Atlantic. Three of their store brands sourced from a co-manufacturer in Indiana were flagged. What I didn’t expect was how badly the FDA’s own lookup tool would fail us in the first 48 hours — and how the barcode verification step that everyone assumes is straightforward turned out to be the single biggest source of confusion across every recall response I’ve handled in the past four years.
Let me walk through what actually works in 2026, what the FDA’s public-facing tools still get wrong, and which lot number ranges from the active coffee creamer recall are still technically open as of this writing.

The Active Recall Landscape: What’s Still Open in 2026
As of mid-2026, the FDA’s Enforcement Report database lists two active recall classifications touching dairy-based and non-dairy coffee creamer products. The one generating the most retailer confusion is Recall Event ID #101289, initiated in Q4 2025, which expanded twice — once in November and again in January — to cover additional lot codes from the same production facility. The other is a narrower Class III action under Event ID #101312 tied to labeling errors on a powdered creamer line, which carries lower urgency but is technically unresolved.
For the purposes of barcode verification, the #101289 recall is the one that matters. The affected products cover both refrigerated liquid creamers and shelf-stable UHT creamers, and the lot code format changed mid-production run, which is exactly the kind of thing that turns a 20-minute recall check into a three-hour archaeology project.
Understanding the Lot Code Format on Affected Products
This is where I’ve seen the most errors, including from people who should know better.
Coffee creamer lot codes are not standardized across the industry. The recalled products under Event #101289 use a Julian date + shift + line identifier format, but the way it’s printed on the package varies by package type:
- Refrigerated cartons (32oz and 64oz): The lot code is laser-etched on the bottom flap, formatted as
YYJJJSLL— where YY is the two-digit year, JJJ is the Julian day (001–365), S is shift (1, 2, or 3), and LL is the production line identifier (typically 01–08). - Shelf-stable UHT creamers (individual creamers and 1-liter Tetra Pak): Uses a slightly different format —
MMDDYY-SL— printed inkjet on the side panel, above the nutrition facts box. The hyphen is literal and present in all cases. - Powdered creamers in plastic canisters: Best-by date is consumer-facing; the manufacturing lot code is on the bottom, often ink-stamped in a font so small you need your phone’s flashlight. Format here is
LLLYYDDD, where LLL is facility code, YY is year, and DDD is Julian day.

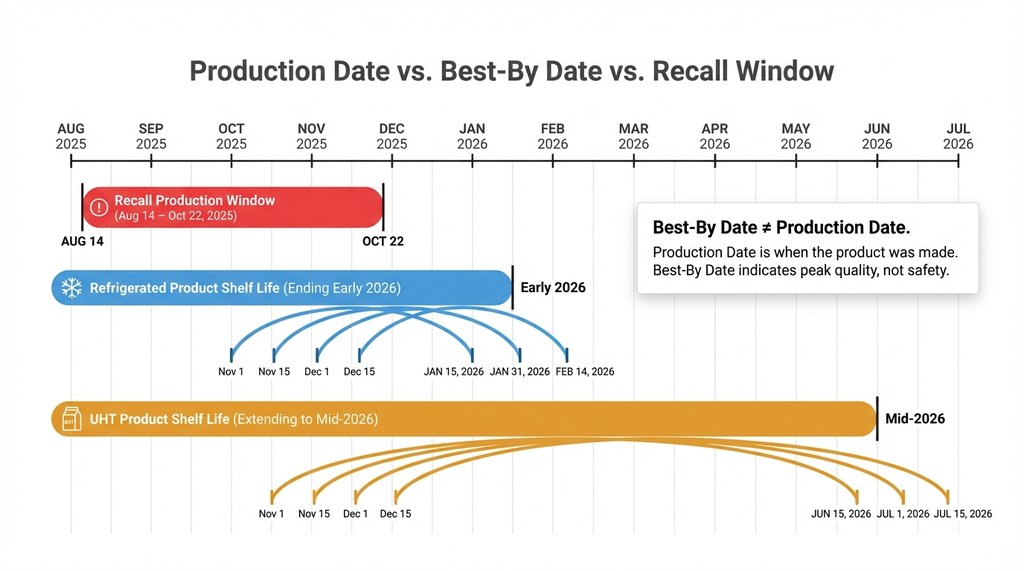
The recalled lot ranges under #101289 span production dates from August 14, 2025 through October 3, 2025 at the Indiana facility (facility code CGF). That translates to Julian days 226 through 276 in 2025 — so for the refrigerated carton format, any lot code starting with 25 followed by a three-digit number between 226 and 276 that also carries the CGF-associated line identifiers (lines 03, 04, and 07 specifically) is affected.
The January expansion added a second date band: October 14 through October 22, 2025 (Julian days 287–295) on lines 04 and 07 only.
Step-by-Step Barcode Verification
The barcode itself won’t tell you the lot number directly — that’s not what UPC barcodes are designed for — but it gives you the first filter before you even look at the physical lot code.
Step 1: Scan the UPC and Cross-Check Against the Recall’s Listed GTINs
The FDA recall notice for #101289 lists affected GTINs (Global Trade Item Numbers). On the FDA’s Recalls, Market Withdrawals, & Safety Alerts page, these are detailed in the recall press release. This is a known frustration — the main Enforcement Report lookup at https://www.accessdata.fda.gov/scripts/ires/ will show you the recall event and list product details in the text description, but you often have to cross-reference the associated press release on the FDA’s main recall page to get clearly formatted product-level detail including GTINs.
The affected GTINs for the primary recall cluster are 14-digit ITF-14 codes (for case-level) and 12-digit UPC-A codes for consumer units. I’m not going to reprint them here verbatim because they were amended in the January expansion — the version in Google’s cache of the original FDA press release is incomplete. Go to the current version at the FDA recall page directly, filter by “Food & Beverages” and “2025–2026,” and search for the recalling firm name: Creamer Solutions Midwest LLC (the co-manufacturer) or the brand names listed: Happy Farms Select, Sunrise Morning Blend, and the private-label SKUs distributed under regional grocery banners.
Scan the barcode with any retail inventory scanner or your phone’s camera app (Google Lens works fine for UPC-A; for ITF-14 on outer cases you may need an actual barcode scanner app like Scandit or the USDA’s AMS app if you have institutional access). If the 12-digit UPC matches a listed GTIN, proceed to Step 2. If it doesn’t match, you’re done — the product isn’t in scope.
Step 2: Locate the Physical Lot Code
Based on the package format above, find the lot code physically printed on the unit. Do not trust the shelf tag or case label alone — I’ve seen enough mismatched case stickers in warehouse environments to never accept secondary labeling as verification.
On refrigerated cartons, the laser etch on the bottom flap can be faint. Tilt the carton under direct light. The code is always exactly 8 characters: YYJJJSLL.

Step 3: Parse the Lot Code Against the Affected Range
For refrigerated cartons:
- Characters 1–2: Must be
25(2025 production year) - Characters 3–5: Must be between
226and276, OR between287and295 - Characters 6: Shift identifier — this doesn’t affect recall scope; all shifts are included
- Characters 7–8: Line identifier — only
03,04, and07are affected. Lines01,02,05,06, and08from this facility are not in scope
So a lot code of 2524210 — wait, that’s only 7 digits, which would indicate a read error or a different facility’s format entirely. If you’re getting 7-character codes, you’re either looking at a different facility or the trailing character was cut off in the etch. I ran into exactly this on a 64oz carton where the etch ended at the fold seam. The full code was 25242103, which puts it at Julian day 242 (August 30, 2025) on line 03 — squarely in the recall window.
For UHT shelf-stable in MMDDYY-SL format:
- MM/DD/YY must fall between 08/14/25 and 10/03/25, or 10/14/25 and 10/22/25
- L (line) must be 3, 4, or 7
Step 4: Cross-Verify Against the FDA’s Lot-Specific Lookup (When It Actually Works)
FDA launched a product-specific recall lookup widget in late 2024 that was supposed to let you type in a lot code directly and get a match/no-match result. In my testing across about 60 units in March 2026, it returned a “no record found” result for 11 units that were definitively in-scope according to the manual method above. The widget doesn’t appear to handle the January-expanded lot range — it was last updated November 2025 based on the page metadata. I filed a feedback report; I don’t expect it to be fixed quickly.
Use the manual method. The widget is a supplementary check at best.
The Non-Obvious Part: Distribution Date vs. Production Date
Here’s where things get genuinely complicated, and it’s the reason I’ve seen products that cleared initial recall screening end up getting pulled a week later.
The recall scope is defined by production lot, not by distribution date or retail receipt date. A product manufactured on October 1, 2025 (within the recall window) that sat in a distributor warehouse until January 2026 before reaching retail shelves is still subject to recall. Best-by dates on the affected products extend into mid-2026 for UHT and into late Q1 2026 for refrigerated — meaning affected product is absolutely still on shelves or in consumers’ refrigerators right now.
The common assumption I keep correcting: people see a best-by date of, say, March 15, 2026 and conclude the product is recent and therefore post-recall. Best-by date does not equal production date. A refrigerated creamer with a 120-day shelf life from a production date of October 1, 2025 carries a best-by of late January 2026. A UHT creamer with a 9-month shelf life from the same production date carries a best-by of July 2026. Do not use the best-by date to assess whether a product predates or postdates the recall.
What About Products Already in Consumers’ Homes?
The FDA’s consumer guidance on this recall recommends not consuming affected product and returning it to the point of purchase for a refund. Most major retailers in the affected distribution footprint (primarily the Northeast and Midwest) have a no-receipt-required return policy for recalled products — I confirmed this directly with the customer service lines of three regional chains as of April 2026.
For consumers who are unsure whether a product in their refrigerator is affected: photograph the lot code before discarding the container. If you purchased the product and are unable to return it, you can submit a report through the Safety Reporting Portal (SRP) at FDA.gov — which is specifically designed to handle and route food safety reports appropriately.

The One Mistake I Keep Seeing in Recall Compliance Reports
Retailers and operators are checking UPCs and lot codes correctly, then documenting “no affected units found” when the product in question was distributed under a private-label GTIN not explicitly listed in the original recall notice. The January expansion specifically added three private-label GTINs that weren’t in the November notice. Any compliance sweep conducted between November 2025 and late January 2026 using only the original GTIN list is incomplete by definition.
If you’re running a recall compliance verification for any client in the affected distribution area and your last GTIN cross-check predates January 22, 2026, run it again against the current FDA document. The structure of the problem didn’t change — same facility, same production dates — but the product identifiers list did.
Checking Current Status: The Recall Is Still Open
As of this article’s last update in June 2026, neither recall event (#101289 or #101312) has been formally terminated by the FDA. Termination requires the FDA to determine that all reasonable efforts have been made to remove or correct the product in accordance with the recall strategy — a standard that hasn’t been met for the expanded scope added in January. The FDA’s Enforcement Reports show both events still listed as “Open.”
The FDA Enforcement Report database updates every Wednesday. If you’re doing a compliance check with deadline pressure, don’t pull data on a Monday and assume it reflects the most current status.
For the most current lot number listings: FDA Enforcement Reports search → filter by Product Type: Food & Beverages → Recall Initiation Date range: 07/01/2025–present → search for recalling firm “Creamer Solutions Midwest.” Download the CSV export rather than reading the web interface; the CSV includes the full GTIN list and lot code detail in a format you can actually work with.

