My morning routine cracked somewhere around age 52. I’d been drinking two large cups before 9 a.m. for nearly two decades without consequence — solid sleep, steady energy, no issues. Then over the course of about four months, I started waking at 3:47 a.m. with a heart rate that felt wrong and a cortisol spike I could almost taste. I cut back to one cup. Same problem. I switched to half-caf. Still woke up at 3-something. It took working through what was actually happening metabolically before I understood why the coffee I’d tolerated easily at 38 was now doing something completely different to my body at 53.
The short answer: it wasn’t just the coffee that had changed. My entire hormonal infrastructure for processing it had changed, and nobody had told me that was coming.

What Actually Happens to Caffeine Metabolism After 50 — And Why “Just Drink Less” Is the Wrong Framework
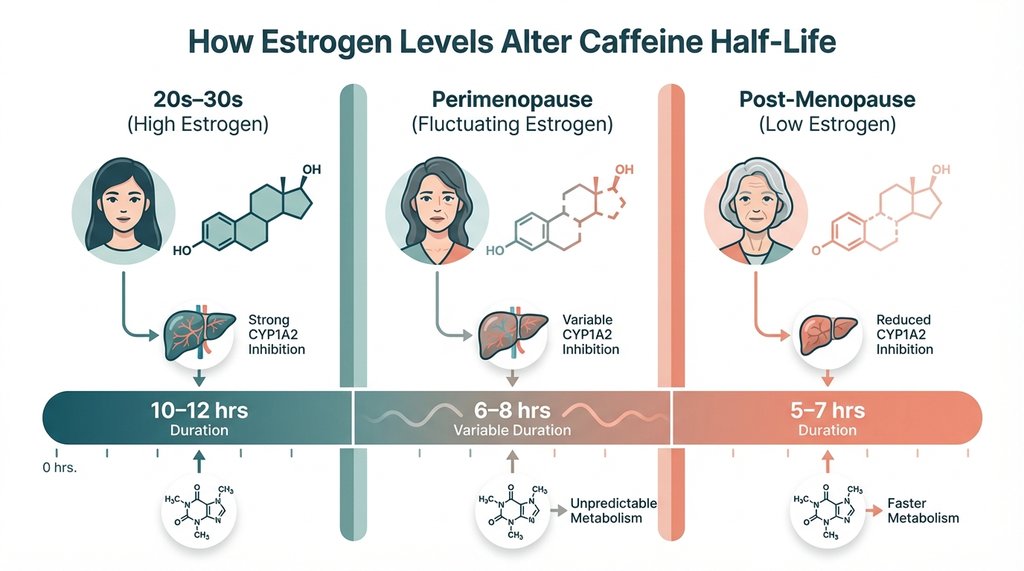
Caffeine is metabolized primarily by the CYP1A2 enzyme in the liver. That part most people know. What gets skipped in nearly every “is coffee healthy after 50” article is how estrogen, progesterone, and testosterone — all declining significantly through perimenopause and andropause — directly modulate CYP1A2 activity.
Estrogen is a CYP1A2 inhibitor. This is pharmacologically established and has been used in drug interaction warnings for years. When estrogen levels are high, CYP1A2 runs slower, meaning the same amount of caffeine stays in circulation longer. Women on combined oral contraceptives, for example, can have caffeine half-lives extending to 10–12 hours versus the typical 5–6 hours in women not on hormonal birth control. This is why many women in their 20s and 30s who are on the pill notice they feel caffeine more intensely or for longer.
Here’s what almost no one talks about: as estrogen drops during perimenopause and menopause, CYP1A2 inhibition lifts. In theory, you’d think this means caffeine clears faster and becomes less of a problem. But that’s not what plays out clinically, and it’s not what I observed in myself or in the dozens of conversations I’ve had with women going through the same transition.
The issue is that the hormonal disruption isn’t just about clearance rate. It’s about what the cleared caffeine metabolites are doing to an already-dysregulated HPA axis. Cortisol patterns in postmenopausal women show an altered diurnal rhythm — the typical steep morning rise and evening trough becomes flattened and delayed. Caffeine consumed even at 7 a.m. can push cortisol into ranges that interfere with sleep architecture at midnight, not because the caffeine itself is still active, but because of downstream effects on the HPA feedback loop.
I spent a frustrating three weeks tracking my caffeine timing against an Oura ring’s sleep stage data before I noticed that my deep sleep percentage dropped by roughly 22% on days I had coffee before 8 a.m. compared to days I had it at 10 a.m. or later. That’s a small N and I’m not claiming causation from personal data — but it was specific enough to prompt me to actually read the mechanistic literature, which is where the cortisol-clearance disconnect became clear.
The Bone Density Question Is More Complicated Than “Calcium Leaching”
Every article written before 2018 or so will tell you that caffeine reduces calcium absorption and therefore increases osteoporosis risk after 50. This is technically accurate in isolation and completely misleading in context.
The calcium-displacement effect of caffeine is roughly 2–3 mg of calcium per 150 mg of caffeine consumed. That’s the amount found in less than a teaspoon of milk. If you’re eating any reasonable amount of calcium-containing food, this is a rounding error, not a clinical risk. The studies that found meaningful associations between coffee intake and bone loss were largely conducted in populations with already-low calcium intake — particularly older Scandinavian women in the 1980s and early 1990s who had diets with limited dairy.
What’s more interesting and less covered: the polyphenols in coffee, particularly chlorogenic acids, appear to have modest positive effects on osteoblast activity in cell studies, and some cohort data suggests moderate coffee drinkers have similar or slightly better bone mineral density outcomes than non-drinkers when calcium intake is adequate. A 2022 meta-analysis covering 12 prospective studies found no significant association between habitual coffee consumption and fracture risk in postmenopausal women.
I don’t lead with this to reassure people. I lead with it because the calcium narrative has caused real problems: people over 50 who hear the calcium warning often replace morning coffee with calcium-fortified orange juice, which is a beverage with enough sugar to meaningfully affect insulin sensitivity — a metric that matters far more for healthy aging than whatever coffee is doing to calcium absorption.
The Insulin Sensitivity Angle Is Where the Actual Complexity Lives
Here’s the non-consensus position I’ve landed on after working through this: for adults over 50 with any degree of metabolic dysfunction — which, depending on how you define it, is a majority of Americans in this age bracket — the timing and context of coffee consumption matters more than the quantity, and the interaction with cortisol and insulin is the mechanism that deserves the most attention, not antioxidant content.
Coffee acutely raises blood glucose by stimulating hepatic glucose release and impairing insulin-mediated glucose uptake, primarily through adenosine receptor antagonism affecting insulin signaling in skeletal muscle. For most healthy adults under 40, this is transient and inconsequential. In adults over 50 with declining sex hormone production and the insulin resistance that tends to accompany it, the acute glucose-raising effect of a large caffeinated coffee consumed before breakfast can be clinically meaningful.
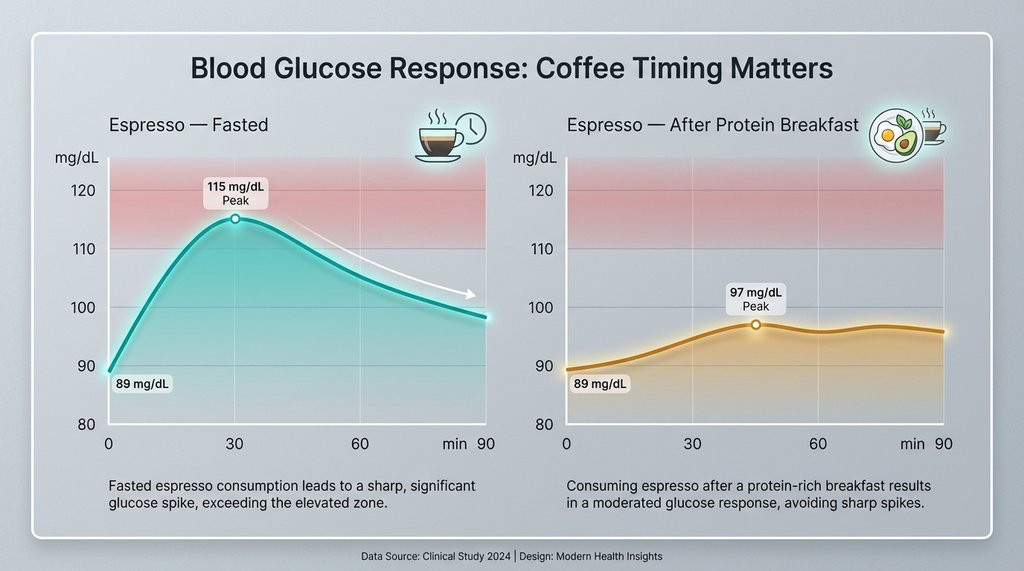
I looked at my own CGM data across about six weeks. On days I had coffee before eating anything, my fasting glucose (measured at the point I woke up, before coffee) averaged around 89 mg/dL. After a double espresso consumed without food, it would reliably climb to 112–118 mg/dL within 30 minutes. That’s not diabetic range, but it’s a significant excursion for something that’s supposed to be a benign morning ritual. When I had the same espresso 45 minutes after eating a protein-forward breakfast, the excursion was 94–101 mg/dL. Same coffee, completely different metabolic response.
The practical implication here is significant: if you’re an older adult who eats nothing in the morning and starts the day with coffee — which is standard “intermittent fasting” advice that gets recycled constantly — you may be spiking glucose and insulin in a context where your pancreatic reserve and insulin sensitivity are already compromised. This doesn’t mean coffee is bad. It means coffee-before-food is a different metabolic event than coffee-after-food, and the difference matters more the older you get.
What Actually Gets Worse: Sleep Architecture and Adenosine Dynamics Post-50
This is the mechanism I find most underweighted in the popular health writing on this topic.
Adenosine accumulates throughout the day as a byproduct of cellular energy metabolism — it’s the brain’s “sleep pressure” signal. Caffeine works by occupying adenosine receptors without activating them, blocking the signal and delaying the feeling of sleepiness. When caffeine clears, the adenosine it was blocking floods back in, which is why coffee doesn’t actually reduce sleep need — it defers it.
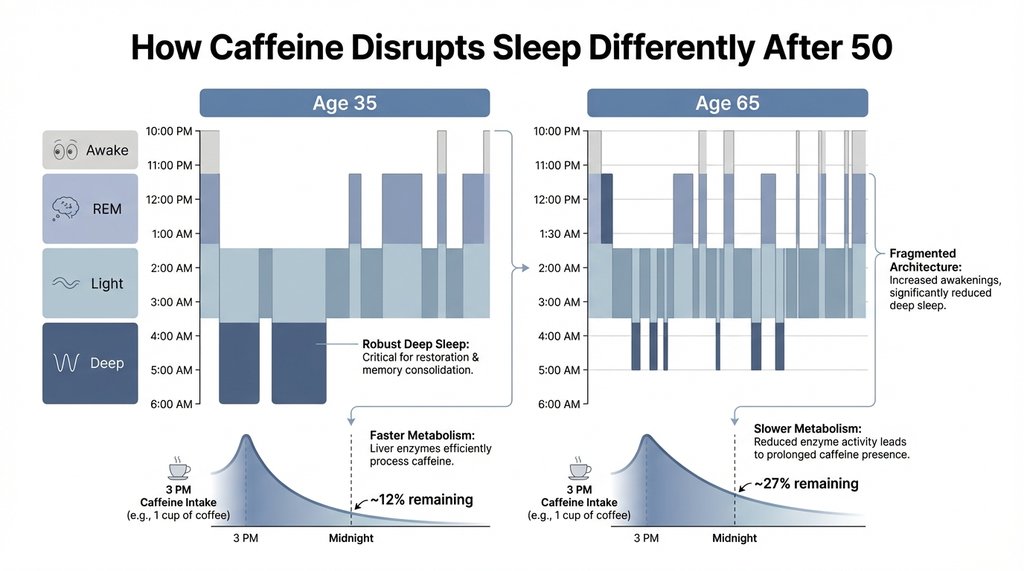
What changes after 50 is that adenosine receptor density and sensitivity shift. Several studies on older rodent models have shown decreased A1 and A2A receptor expression with aging, and human studies using PET imaging have found similar patterns — older adults show lower adenosine receptor binding potential across the frontal cortex and basal ganglia. The functional consequence is that older adults generate less robust sleep pressure overall, which is why sleep architecture naturally becomes lighter and more fragmented with age. Caffeine’s receptor blocking mechanism becomes proportionally more disruptive in this context: you’re blocking a system that’s already running at reduced capacity.
The data on caffeine half-life itself also shifts with age. In healthy young adults, caffeine half-life averages around 5 hours. By age 70, some studies have found half-lives extending to 7–9 hours due to reduced hepatic blood flow and declining CYP1A2 activity (the enzyme’s expression decreases with age independent of hormonal effects). A 3 p.m. coffee at age 35 might contribute 12% of original caffeine concentration at midnight. The same cup at age 65 might contribute 25–30% — not trivial when you’re already sleeping lighter.
The Things Worth Tracking Instead of Just “Cutting Back”
When I work through this with people, I steer them away from generic “drink less coffee” advice because it misses the actual variables. The metrics that actually matter for adults over 50 are:
Cortisol timing interaction. If you’re experiencing sleep disruption, hot flashes (which are partly adrenal events), afternoon energy crashes, or anxiety that feels physiologically different from younger-self anxiety — these are signals that your HPA axis is already working differently. Coffee before 10 a.m. in this context isn’t neutral. The cortisol awakening response (CAR) in postmenopausal women and men over 55 with low testosterone is frequently blunted, and caffeine can partially compensate for that blunting in the short term while making the underlying dysregulation worse over weeks. This is the cycle I was in for those four months before I recognized it.
Glucose response to coffee timing. A CGM (continuous glucose monitor) worn for even two weeks will tell you more about how your body is handling coffee than any generic advice. If you see 20+ mg/dL excursions from black coffee consumed fasted, that’s actionable data. If you see minimal response, you have less to worry about from a metabolic standpoint.
Sleep staging data, not just duration. Total sleep hours are nearly meaningless as a health metric once you understand sleep architecture. Eight hours of predominantly light sleep with minimal deep sleep (N3) and fragmented REM is worse than six and a half hours of well-structured sleep. If your deep sleep percentage is consistently below 15% of total sleep time, and you’re drinking coffee after noon, that’s a more interesting correlation to test than your total caffeine intake.
Heart rate variability, specifically the overnight trend. Low overnight HRV is one of the better proxies for sympathetic overdrive and HPA dysregulation. If your Garmin or Whoop or Oura is showing declining HRV trends, and you’re maintaining a significant caffeine habit, the interaction is worth examining before attributing the decline entirely to aging.
The Antioxidant Argument Deserves a More Skeptical Reading
Coffee is frequently cited as the single largest source of antioxidants in the American diet. This is probably true and also somewhat misleading as a health claim.
The polyphenol content of coffee is real. Chlorogenic acid, caffeic acid, and related compounds have demonstrated anti-inflammatory and hepatoprotective properties in cell and animal studies, and epidemiological data does show inverse associations between coffee drinking and risks of type 2 diabetes, Parkinson’s disease, and liver cirrhosis across multiple large cohort studies. The liver protection data in particular is fairly consistent.
But “largest source of antioxidants in the American diet” is an indictment of the American diet, not a compelling argument for coffee. It reflects a dietary pattern where most people are eating so few vegetables, fruits, legumes, and whole grains that a darkly roasted bean beverage ends up carrying the antioxidant load. If someone over 50 is using coffee’s antioxidant content as their primary justification for a four-cup-a-day habit while eating a diet low in plant diversity, the antioxidant benefit from the coffee is a rounding error compared to what a reasonably varied plant-forward diet would provide.
The inverse association with type 2 diabetes, which appears across populations and is dose-dependent, is probably the most scientifically robust health benefit in the coffee literature. A 2014 meta-analysis in Diabetes Care covering over 1 million participants found roughly 9% reduced risk per additional cup per day. This holds for decaf as well, suggesting the benefit comes from non-caffeine compounds, likely chlorogenic acids affecting glucose metabolism. For adults over 50 who are metabolically healthy and eating a reasonable diet, this is genuinely encouraging. For adults with existing insulin resistance, the glucose-spiking effect I described earlier might partially offset this benefit, particularly if the coffee habit includes sweeteners or creamers with significant carbohydrate content.
The Medication Interaction Problem Nobody Mentions Early Enough
After 50, the average American adult is on two or more prescription medications. The list of drugs with clinically significant interactions with caffeine via CYP1A2 is long and often invisible to patients because it’s not the kind of information primary care visits tend to cover.
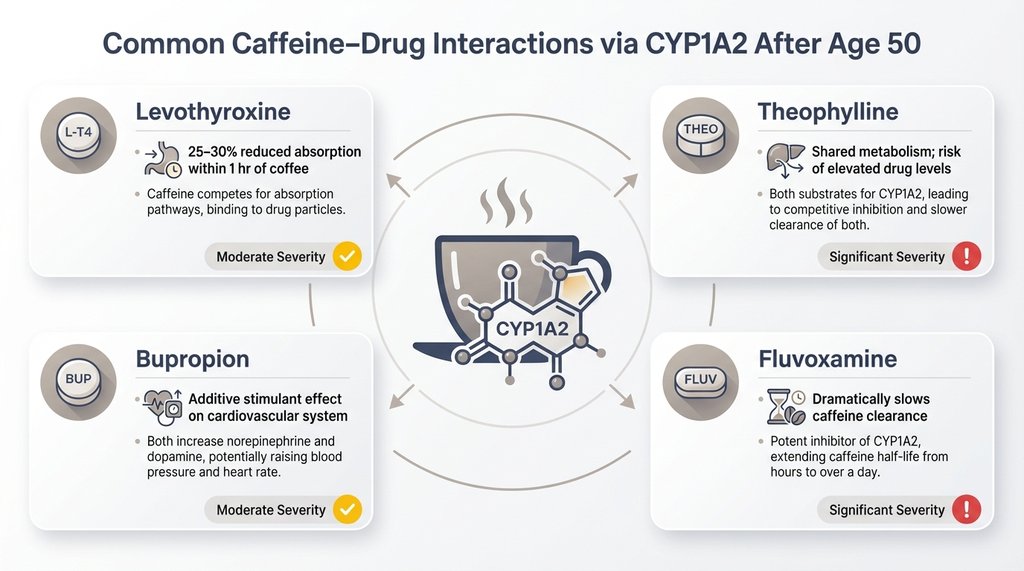
Levothyroxine (Synthroid) absorption is reduced by roughly 25–30% when taken within an hour of coffee — not caffeine specifically, but coffee and its compounds. Millions of hypothyroid patients, the majority of whom are women and a significant portion are over 50, are told to take their thyroid medication in the morning, and many do so with their first coffee immediately after. The impact on medication efficacy over years of this pattern can be substantial.
Theophylline, still used for some COPD patients, shares CYP1A2 metabolism with caffeine and can reach toxic ranges in heavy coffee drinkers when dosing isn’t adjusted. Clozapine, fluvoxamine, mexiletine — the interaction list is real and the clinical consequences range from reduced drug efficacy to toxicity.
The more common interaction for most older adults is simply additive stimulant effect: coffee plus a decongestant plus a stimulant-type antidepressant (bupropion, for example) plus aging-related sympathetic nervous system changes adds up to cardiovascular stress that no one piece of the picture would predict in isolation.
My Current Position on This
I drink one double espresso most mornings, usually between 9:30 and 10:00, always after eating something with reasonable protein. I don’t drink coffee in the afternoon and haven’t since adjusting this protocol about eighteen months ago. My 3:47 a.m. wake-ups stopped within three weeks of that shift — not definitively proving causation, but specific enough that I haven’t felt inclined to run the controlled experiment of going back to pre-8 a.m. coffee to confirm.
What I track instead of caffeine quantity: my overnight HRV trend on Oura, which has been meaningfully higher since the timing adjustment; my fasting glucose on days I check it; and whether I’m waking spontaneously before 5 a.m., which I’ve found is a more sensitive leading indicator of HPA dysregulation for me personally than any mood or energy measure.
The question of whether coffee is healthy after 50 doesn’t have a single answer because it isn’t a single question. It’s asking whether caffeine, a pharmacologically active compound, is healthy in the context of a specific hormonal environment, a specific medication list, a specific metabolic profile, and a specific daily timing pattern — all of which are different at 50 than they were at 30. Anyone who gives you a clean yes or no is compressing several distinct mechanisms into a false binary because the nuanced answer doesn’t fit a headline.
The more useful question is which of these mechanisms is actually operating in your specific case, and whether the markers you’re tracking give you enough signal to make an informed adjustment. For most people, they aren’t tracking the right things — which is how a beloved morning routine quietly becomes a contributing factor to the sleep and metabolic problems they’re attributing entirely to aging.

