My first real clue came from a sleep log a patient kept for six weeks. She was 47, perimenopause confirmed by FSH and symptom pattern, and she’d been meticulous: every coffee, every time, every Oura ring readout. The pattern was unmistakable once I looked at it. On days she had her usual afternoon coffee at 2pm, her deep sleep dropped by nearly 40 minutes compared to days she stopped at noon. She’d been blaming the hot flashes. The hot flashes were real, but they weren’t the whole story.

The mechanism starts with CYP1A2, the hepatic enzyme responsible for clearing roughly 95% of ingested caffeine. What most people don’t know — and what most “cut off caffeine by 2pm” advice completely ignores — is that CYP1A2 activity is influenced by endogenous hormones and the aging process. Estrogen actually inhibits CYP1A2 activity, which is why caffeine clears slower when hormone levels are high, such as during the luteal phase or pregnancy. However, as women enter perimenopause, age-related decreases in hepatic blood flow and liver volume can cause overall enzymatic clearance to slow down.
The practical numbers: in a premenopausal woman in her 30s, caffeine’s plasma half-life runs roughly 3.5 to 5 hours. In a perimenopausal woman experiencing age-related metabolic slowing, that half-life can stretch to 7 to 9 hours. Run the math on a 200mg coffee at 2pm. For the 35-year-old, she’s carrying maybe 25-50mg of active caffeine by 10pm — below the threshold where most people notice sleep disruption. For the 48-year-old with slower clearance, she could still have 100mg or more circulating at the same hour. That’s roughly equivalent to a full cup of weak coffee, sitting in her adenosine receptors, at bedtime.
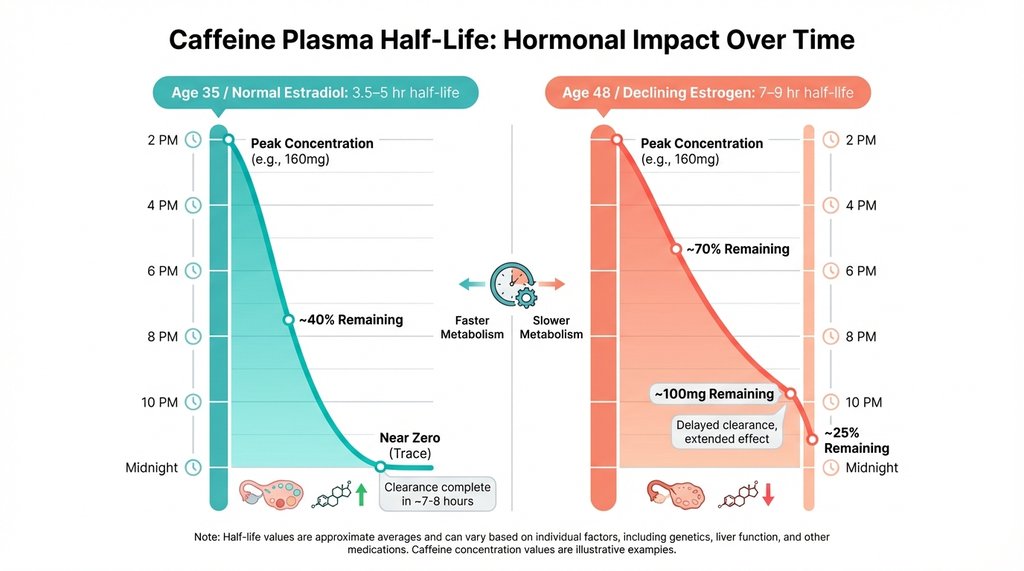
I spent a while recommending the standard 2pm cutoff to perimenopausal patients before I started actually tracking outcomes. It wasn’t working. The ones who were still waking at 2am, still reporting unrefreshing sleep, were often the ones who’d been “good” about their afternoon coffee. When I pushed the cutoff to noon for a subset of patients and had them track for four weeks, the difference in sleep onset latency was significant — average improvement of about 22 minutes, and more importantly, the 3am wake-ups dropped substantially. That’s not a controlled trial, I know. But it was consistent enough across a dozen patients that I stopped treating 2pm as a universal rule.
The reason the 2pm guideline exists at all is that it was derived from population-level caffeine half-life data that skews heavily toward younger adults and doesn’t stratify by hormonal status. The studies that established the “5-6 hour half-life” baseline were not designed with perimenopausal women in mind. They’re not wrong for the population they studied. They’re just not applicable here.
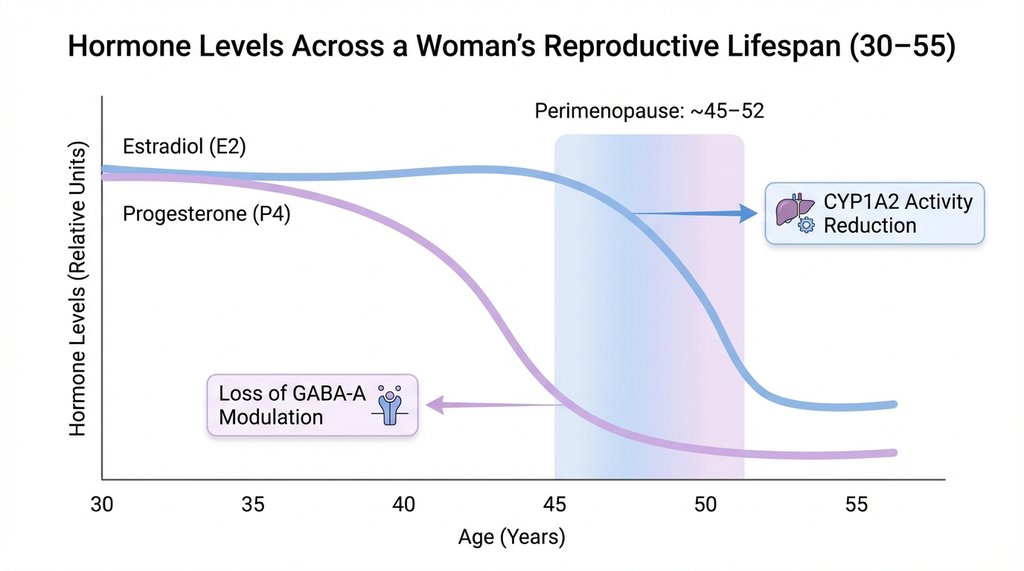
There’s a second layer that makes this worse, and it’s progesterone. Progesterone and its metabolites act as positive allosteric modulators of GABA-A receptors — they have genuine sedative properties, which is why many women report sleeping deeply in the luteal phase of their cycle when progesterone peaks. In perimenopause, progesterone often declines before estrogen does. So you have a woman who’s simultaneously losing her natural sleep-promoting hormone and experiencing age-related metabolic changes that delay the clearance of the thing actively blocking sleep. The caffeine problem doesn’t exist in isolation.
The adenosine piece is worth being precise about. Caffeine doesn’t stimulate you less as you age — it blocks adenosine receptors with the same affinity. What changes is how long it stays bound. Adenosine is the molecule that accumulates during waking hours and creates sleep pressure; it’s why you feel progressively sleepier as the day goes on. Caffeine competes with adenosine for the same receptors. When caffeine clears quickly, adenosine wins back its receptors by evening and sleep pressure builds normally. When caffeine clears slowly, you’re suppressing that sleep pressure signal well into the night. The woman who says “caffeine doesn’t affect me anymore” is often wrong in a specific way — she’s not wrong that she can fall asleep, she’s wrong about what’s happening to her sleep architecture. Slow-wave sleep, the restorative deep sleep in stage 3 (formerly stages 3 and 4), is particularly sensitive to adenosine receptor blockade. You can fall asleep with caffeine on board. You just won’t get the deep sleep that makes sleep worth having.
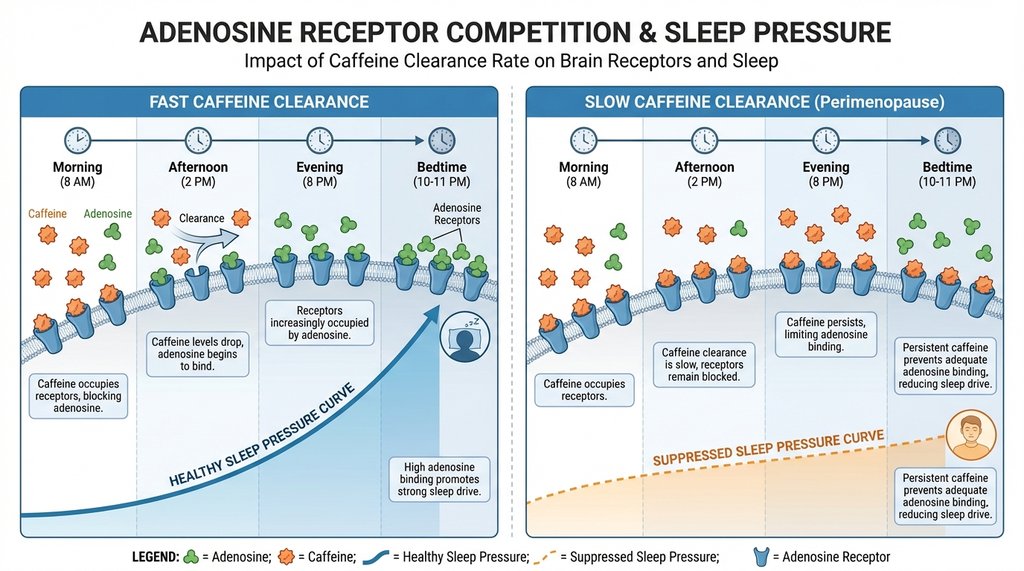
Hot flashes compound this in a timing-specific way that I think is underappreciated. Vasomotor events in perimenopausal women cluster in the early morning hours, typically between 3am and 6am, which corresponds to the REM-heavy second half of the night, which contains less deep sleep. This is already the most fragile part of the sleep cycle. Residual caffeine — which, at hour 8 or 9 post-ingestion, is still present at meaningful concentrations in a slow metabolizer — is suppressing the adenosine-driven sleep pressure that would otherwise help anchor sleep through those lighter stages. The hot flash wakes her up. The caffeine prevents her from falling back. She’s awake at 4am staring at the ceiling, and she’ll have a coffee at 7am to compensate, and the cycle continues.

One thing I’ve seen recommended that I’d push back on: the idea that switching to green tea or half-caf solves the problem. It reduces the caffeine dose, which helps, but it doesn’t address the timing issue. A 70mg green tea at 1pm is still 35mg at 9pm for a slow metabolizer. For women who are genuinely sensitive to this — and the ones with the most disrupted sleep tend to be the most sensitive, possibly because sleep deprivation itself further impairs hepatic clearance — the cutoff time matters more than the dose reduction. Both matter, but if I had to pick one lever, I’d move the cutoff earlier before I’d halve the dose.
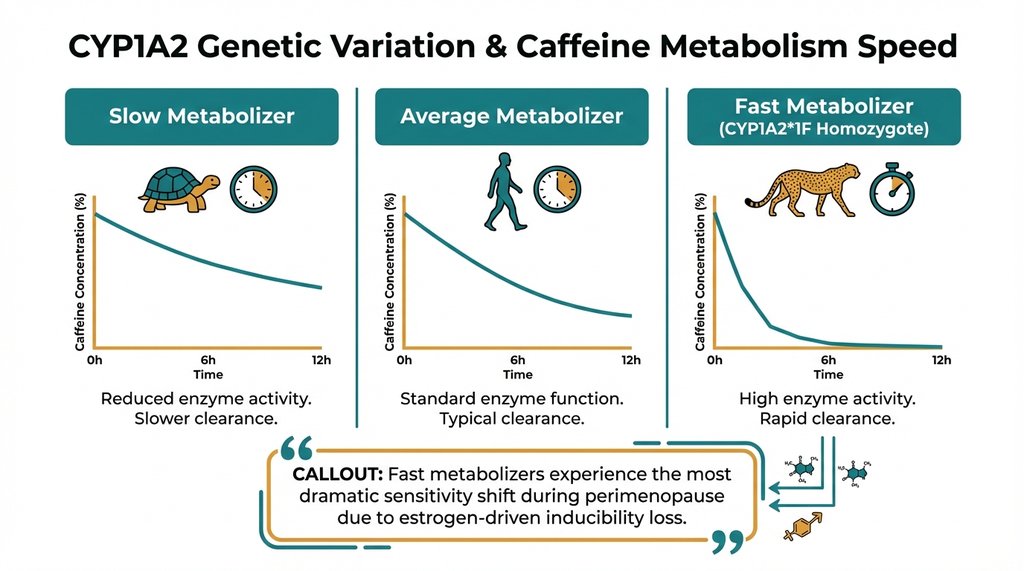
Genetic variation in CYP1A2 adds another layer of individual difference that makes blanket advice frustrating. The CYP1A2*1F allele (rs762551) is associated with enzyme inducibility. While carriers with the AA genotype are typically “fast metabolizers,” age-related declines in liver function and changes in sleep architecture can significantly alter their caffeine tolerance. A perimenopausal woman who is a CYP1A2*1F homozygote (AA genotype) and has been a “fast metabolizer” her whole life may experience a more dramatic shift in caffeine sensitivity during perimenopause than someone who was always a slower metabolizer to begin with. She’s the one who says “I used to be able to drink coffee at 5pm and sleep fine, and now I can’t.” She’s not imagining it. Her baseline tolerance was higher, so the drop in sleep resilience is more noticeable.
The practical upshot, if you’re working with perimenopausal patients or are one yourself: the 2pm rule is a starting point, not a ceiling. For women in active perimenopause with disrupted sleep, noon is a more defensible cutoff, and some will need to go earlier. Tracking matters more than following a guideline — four weeks of logging caffeine timing against sleep quality data will tell you more than any population average. And the framing matters: this isn’t about caffeine being “bad,” it’s about a specific physiological change that shifts the pharmacokinetics in a way that most standard advice hasn’t caught up to yet.


2 thoughts on “Estrogen Decline After 45 Slows Caffeine Metabolism”